Textbook Question
Draw the structures of the following compounds.
c. 3,3-diethyl-4-(2,2-dimethylpropyl)octane
3
views

 Verified step by step guidance
Verified step by step guidance
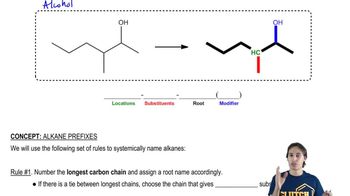

Draw the structures of the following compounds.
c. 3,3-diethyl-4-(2,2-dimethylpropyl)octane
List each set of compounds in order of increasing boiling point.
b. Octane, (CH3)3C—C(CH3)3 and CH3CH2C(CH3)2CH2CH2CH3
List each set of compounds in order of increasing boiling point.
a. hexane, octane, and decane
Draw Newman projections of the following molecules viewed from the direction of the blue arrows.
(a)
Draw the structures of the following compounds.
a. 4-(1,1-dimethylethyl)octane
Draw the structures of the following compounds.
b. 5-(1,2,2-trimethylpropyl)nonane