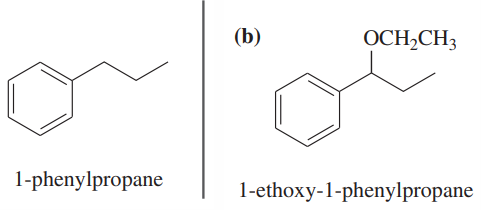
Show how you would convert (in one or two steps) 1-phenylpropane to the three products shown below. In each case, explain what unwanted reactions might produce undesirable impurities in the product.

Show how you would convert (in one or two steps) 1-phenylpropane to the three products shown below. In each case, explain what unwanted reactions might produce undesirable impurities in the product.
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Nucleophilic Substitution
Rearrangement Reactions

Side Reactions and Impurities
Show how you would prepare cyclopentene from each compound.
c. cyclopentane (not by dehydrogenation)
Propose mechanisms for the following reactions. Additional products may be formed, but your mechanism only needs to explain the products shown.
(a)
E1 eliminations of alkyl halides are rarely useful for synthetic purposes because they give mixtures of substitution and elimination products. Explain why the sulfuric acid-catalyzed dehydration of cyclohexanol gives a good yield of cyclohexene even though the reaction goes by an E1 mechanism. (Hint: What are the nucleophiles in the reaction mixture? What products are formed if these nucleophiles attack the carbocation? What further reactions can these substitution products undergo?)
Show how you would convert (in one or two steps) 1-phenylpropane to the three products shown below. In each case, explain what unwanted reactions might produce undesirable impurities in the product.
Show how you would prepare cyclopentene from each compound.
a. cyclopentanol
b. cyclopentyl bromide
