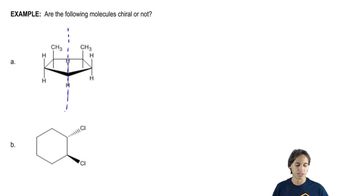
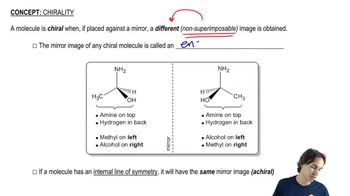
Textbook Question
For each of the compounds described by the following names,1. draw a three-dimensional representation.2. star (*) each chiral center.3. draw any planes of symmetry.4. draw any enantiomer.5. draw any diastereomers.6. label each structure you have drawn as chiral or achiral.c. (2R,3S)-2,3-dibromohexaned. (1R,2R)-1,2-dibromocyclohexane