Show how you would convert propan-1-ol to the following compounds using tosylate intermediates. You may use whatever additional reagents are needed.
c. CH3CH2CH2OCH2CH3, ethyl proyl ether
d. CH3CH2CH2CN, butyronitrile

 Verified step by step guidance
Verified step by step guidance
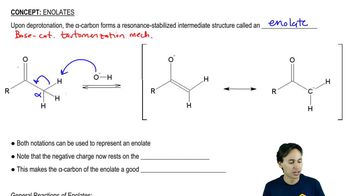

Show how you would convert propan-1-ol to the following compounds using tosylate intermediates. You may use whatever additional reagents are needed.
c. CH3CH2CH2OCH2CH3, ethyl proyl ether
d. CH3CH2CH2CN, butyronitrile
A chronic alcoholic requires a much larger dose of ethanol as an antidote to methanol poisoning than does a nonalcoholic patient. Suggest a reason why a larger dose of the competitive inhibitor is required for an alcoholic.
Predict the major products of the following reactions.
(d) the tosylate of cyclohexylmethanol + excess NH3
(e) n-butyl tosylate + sodium acetylide, H–C≡C:– +Na
Predict the products of the following reactions.
(c) 1-methylcyclohexanol + H2SO4, heat
(d) product of (c) + H2, Pt
Predict the products of the following reactions.
(a) cyclohexylmethanol + TsCl/pyridine
(b) product of (a) + LiAlH4
Predict the major products of the following reactions.
(a) ethyl tosylate + potassium tert-butoxide
(b) isobutyl tosylate + NaI
(c) (R)-2-hexyl tosylate + NaCN