Textbook Question
For which of the following reactions would you expect elimination to be more favored than substitution?
(e)
1
views

 Mullins 1st Edition
Mullins 1st Edition Ch. 12 - Substitution and Elimination: Reactions of Haloalkanes
Ch. 12 - Substitution and Elimination: Reactions of Haloalkanes Problem 50b
Problem 50b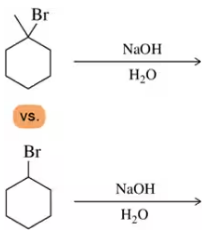
 Verified step by step guidance
Verified step by step guidance


For which of the following reactions would you expect elimination to be more favored than substitution?
(e)
For the following reactions we have not seen yet, which side, if either, would be favored by increasing the temperature?
(c)
For which of the following reactions would you expect elimination to be more favored than substitution?
(a)
For the following reactions we have not seen yet, which side, if either, would be favored by increasing the temperature?
(d)
For which of the following reactions would you expect elimination to be more favored than substitution?
(d)
For which of the following reactions would you expect elimination to be more favored than substitution?
(c)