Answer the following questions for the MOs of 1,3-butadiene:
a. Which are bonding MOs, and which are * antibonding MOs?
b. Which MOs are symmetric, and which are antisymmetric?
c. Which MO is the HOMO and which is the LUMO in the ground state?

 Bruice 8th Edition
Bruice 8th Edition Ch. 8 - Delocalized Electrons: Their Effect on Stability, pKa, and the Products of a Reaction • Aromaticity and Electronic Effects: An Introduction to the Reactions of Benzene
Ch. 8 - Delocalized Electrons: Their Effect on Stability, pKa, and the Products of a Reaction • Aromaticity and Electronic Effects: An Introduction to the Reactions of Benzene Problem 14
Problem 14 Verified step by step guidance
Verified step by step guidance
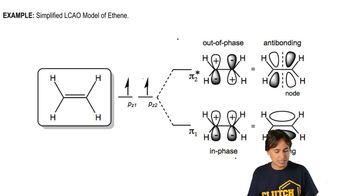

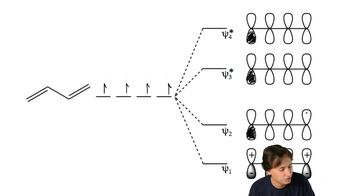
Answer the following questions for the MOs of 1,3-butadiene:
a. Which are bonding MOs, and which are * antibonding MOs?
b. Which MOs are symmetric, and which are antisymmetric?
c. Which MO is the HOMO and which is the LUMO in the ground state?
a. Draw resonance contributors for the following species. Do not include structures that are so unstable that their contributions to the resonance hybrid would be negligible. Indicate which are major contributors and which are minor contributors to the resonance hybrid.
b. Do any of the species have resonance contributors that all contribute equally to the resonance hybrid?
11.
Answer the following questions for the MOs of 1,3-butadiene:
d. Which MO is the HOMO and which is the LUMO in the excited state?
e. What is the relationship between the HOMO and the LUMO and symmetric and antisymmetric orbitals?
Which member of each pair is the stronger base?
a. ethylamine or aniline
b. ethylamine or ethoxide ion
Which has the greater delocalization energy?
Name the following dienes and rank them from most stable to least stable.