Textbook Question
Identify the group number in the periodic table of X, a representative element, in each of the following ionic compounds:
a. XCl3

 Verified step by step guidance
Verified step by step guidance
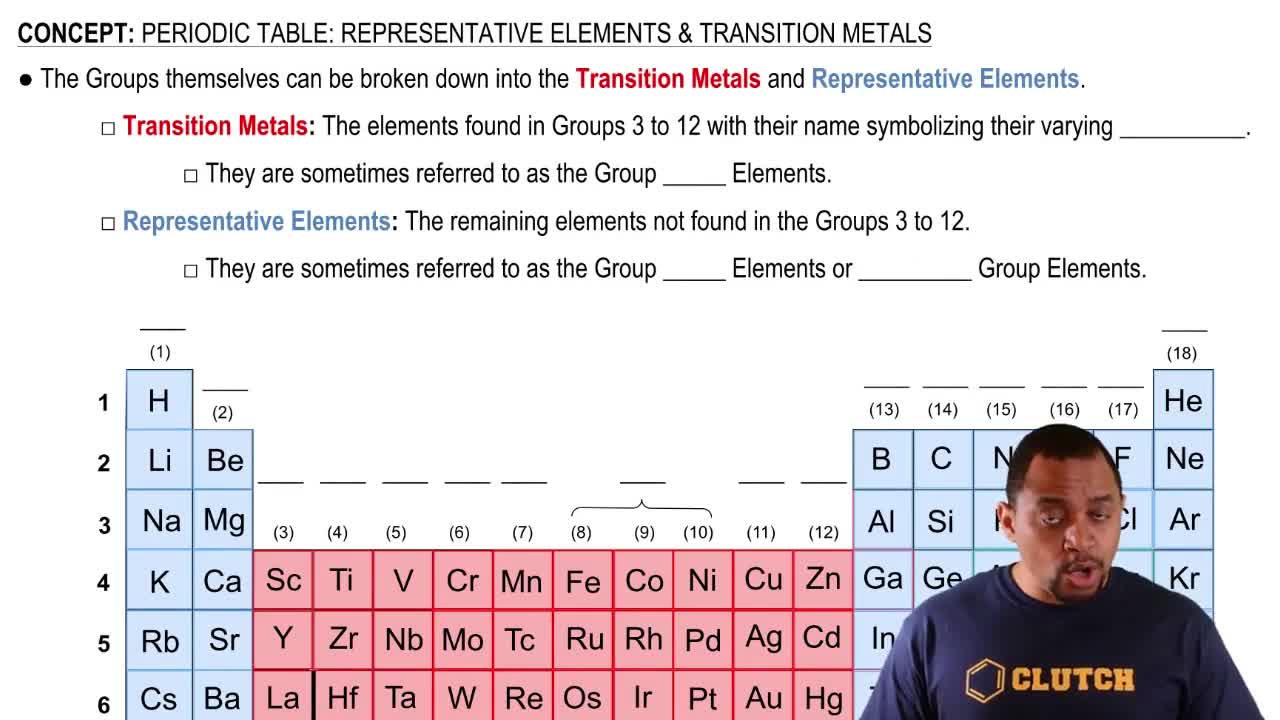


Identify the group number in the periodic table of X, a representative element, in each of the following ionic compounds:
a. XCl3
Classify each of the following as ionic or molecular, and name each:
b. Cl2O7
Identify the group number in the periodic table of X, a representative element, in each of the following ionic compounds:
b. Al2X3
Complete the Lewis structure for each of the following:
a.
Classify each of the following as ionic or molecular, and name each:
a. Li2HPO4
Predict the shape and polarity of each of the following molecules, which have polar covalent bonds:
a. A central atom with three identical bonded atoms and one lone pair.