
Calculate 3OH-4 for each of the following solutions, and indicate whether the solution is acidic, basic, or neutral: (b) 3H+4 = 2.5 * 10-10 M
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
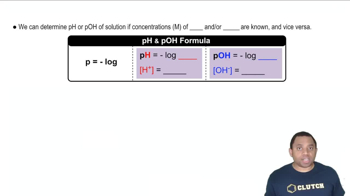
pH and pOH
Acidic, Basic, and Neutral Solutions

Calculating pOH from H+ Concentration

Deuterium oxide 1D2O, where D is deuterium, the hydrogen-2 isotope) has an ion-product constant, Kw, of 8.9 * 10-16 at 20 °C. Calculate 3D+4 and 3OD-4 for pure (neutral) D2O at this temperature.
Write the expression for the ion product constant for water, Kw.
Calculate 3OH-4 for each of the following solutions, and indicate whether the solution is acidic, basic, or neutral: (a) 3H+4 = 0.0505 M
Calculate 3OH-4 for each of the following solutions, and indicate whether the solution is acidic, basic, or neutral: (c) a solution in which 3H+4 is 1000 times greater than 3OH-4.
Calculate [H+] for each of the following solutions, and indicate whether the solution is acidic, basic, or neutral: (a) [OH-] = 0.00045 M (b) [OH-] = 8.8 × 10-9 M (c) a solution in which [OH-] is 100 times greater than [H+].
